|
Chloride, on the other hand, has an electron configuration of 1s2 2s2 2p6 3s2 3p5. 
Since electrons are negatively charged, losing one results in a positive ion with a charge of +1. It can achieve a full outer shell by losing one electron from its 3s sub-shell, resulting in a configuration of 1s2 2s2 2p6. Sodium has an electron configuration of 1s2 2s2 2p6 3s1. We can determine the charges of these ions based on their electron configurations. Sodium chloride is a compound made up of positive sodium cations and negative chloride anions. 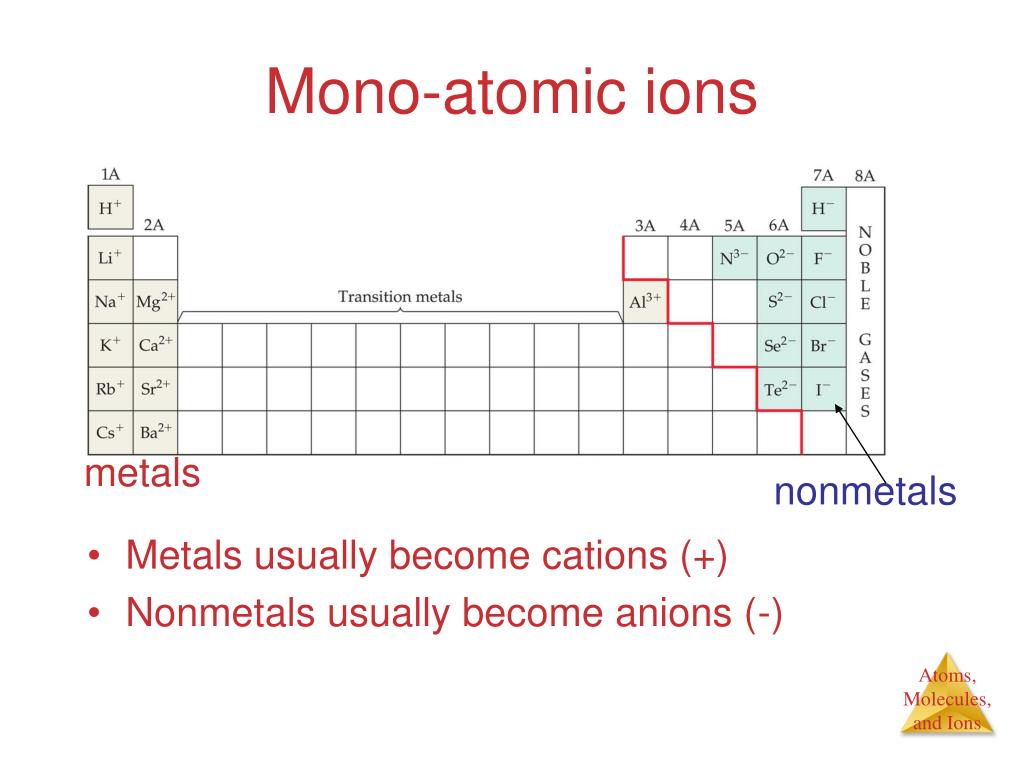
Now, let's consider an example of ionic bonding. When mixed together, cations and anions are electrostatically attracted to one another, and ionic bonding is simply another term for this attraction. This is known as electrostatic attraction you might also remember that this is the force that attracts electrons towards the nucleus in an atom. When two oppositely charged species are close by, they attract each other. Electrostatic attractionįorming ions is only half the picture - By definition, ionic bonding doesn’t involve the transfer of electrons at all! Rather, it is about the interaction between these ions as a result of gaining or losing electrons. Overall, the charges on the ions cancel out, making a neutral compound.We call this transfer of electrons electrovalence. This results in an anion.Both ions end up with full outer shells of electrons.In ionic bonding, the cation is always a metal and the anion is always a non-metal. Because electrons are negative, this results in a cation.The other element gains these electrons. In both cases, the ions have the electron configuration of a noble gas. Ionic bonding always occurs between positively charged ions, called cations, and negatively charged ions, called anions. Ions are atoms that have gained or lost one or more electrons to form a charged particle. So, let's dive in and learn about it! Ions Understanding ionic bonding is important in understanding how atoms interact with each other. Let's explore this process a little further. Oppositely charged ions bond ionically with each other by electrostatic attraction. One species loses electrons, and the other gains them, forming ions. But when a metal and a non-metal come together, the easiest way for both to obtain a full outer shell is by transferring electrons. Groups of metal atoms of the same element will lose electrons to form positive ions in a sea of delocalized electrons (see Metallic Bonding). Non-metals can share their outer shell electrons by joining up in pairs, trios, or larger groups (see Covalent Bond). So, let's dive in and learn about it! Ionic bonding definitionĪtoms can achieve a noble gas structure in different ways. Finally, we'll review the evidence for ionic bonding. We'll also talk about the strength of ionic bonding and ionic radius. We'll explain what ionic bonding is, show examples and diagrams of common ionic compounds, and explore giant ionic lattices and their properties. This article will talk about ionic bonding in physical chemistry. Ionic bonding is a way of achieving this stable configuration. However, other elements don't have this stable arrangement and must gain, lose, or share electrons. They have a full outer shell of electrons, which is the most stable electron configuration. Group 8 elements, also called noble gases, are known for not reacting with other elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
